Early actionability
Precision decision-making when time, space, and certainty are in short supply


A new standard in accurate, actionable diagnostics for better, faster decision-making in the ED.

Watch the video to hear directly from Tim Sweeney, Co-Founder and CEO of Inflammatix.

Voice of
Tim Sweeney, MD, PhD
The TriVerity Test provides three actionable scores giving the likelihood of:
*Defined as the risk of progression to needing mechanical ventilation, vasopressors, and/or renal replacement therapy (RRT) within seven days.
THE SCIENCE BEHIND TRIVERITY
Inflammatix host response diagnostics are powered by ‘reading’ the immune system. Our immune system has developed targeted responses to different types of infections and other diseases over millions of years of evolution.
Leveraging our expertise in bioinformatics, including machine learning and artificial intelligence, and molecular biology, we are harnessing the ability to interpret the immune response to develop tests aimed to help physicians make confident and timely clinical decisions.
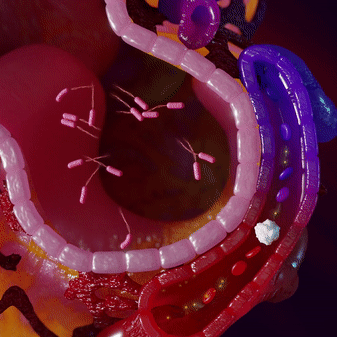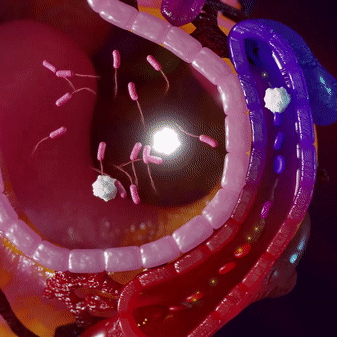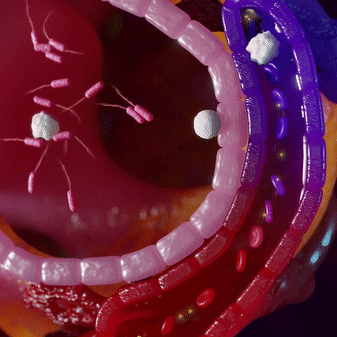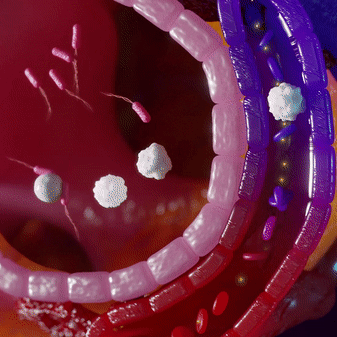
Most infections are locally contained and do not penetrate into the bloodstream. This makes pathogen-targeting tests insensitive for infections. Immune cells fight the infection locally and then circulate into the blood, where we can measure their ‘signatures’ using mRNA profiles.
1. Liesenfeld O, Arora S, Aufderheide T, et al. Rapid and Accurate Diagnosis and Prognosis of Acute Infections and Sepsis from Whole Blood Using Host Response mRNA Amplification and Machine-Learning Classifiers. Preprint published October 10, 2024. Available at: https://doi.org/10.21203/rs.3.rs-5194992/v1