COVID-19 and Sepsis: Risk-Stratifying Patients Based on Their Immune Response
Host-response diagnostics that “read” the immune response can help clinicians make better, faster treatment decisions
Even before the pandemic, about 15 million patients per year presented to US emergency rooms with suspected acute infections or sepsis. Physicians often struggle with rapid and accurate diagnosis and risk stratification in these patients, as symptoms are often nonspecific and similar to noninfectious causes of illness.
The COVID-19 pandemic has shone a spotlight on sepsis, and on the current diagnostic challenges facing those working in and supporting the emergency setting. Challenges include how we determine the presence of infection, whether it is bacterial or viral in origin, and importantly, how we risk-stratify patients. More than ever, we need severely ill patients to be admitted and treated quickly and low-risk patients sent home to recover, preserving valuable hospital resources.

Missed infections
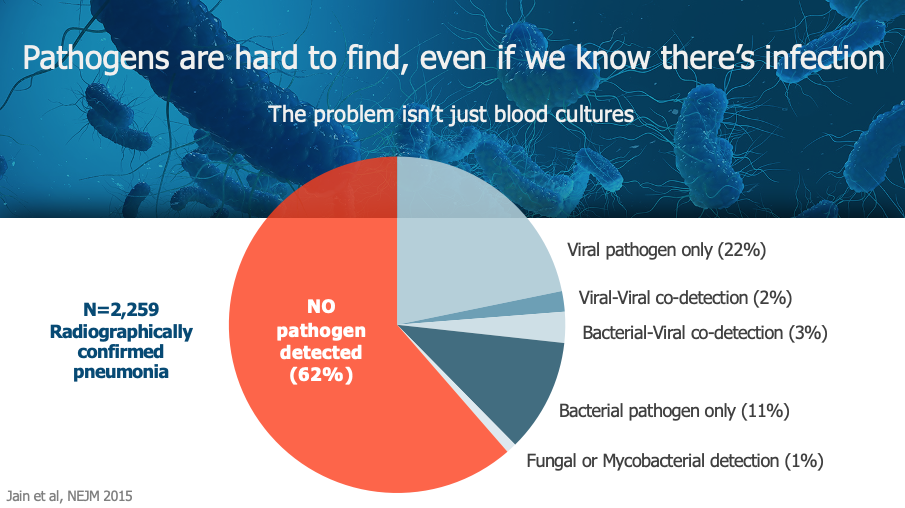
Most infections aren’t found in the bloodstream and are currently missed by standard of care labs. Pathogens are difficult to find and testing is slow even when we know they are there. In fact, in a large New England Journal of Medicine case study, patients with radiographically confirmed pneumonia turned up no pathogens 62 percent of the time after their doctors ran blood, nasal, and sputum cultures. Biomarker testing may be faster, but commonly measured markers such as C-reactive protein (CRP) and procalcitonin (PCT) are nonspecific for infection and not accurate enough to be actionable in a busy emergency room.
The ongoing COVID-19 pandemic has only compounded the need for rapid and accurate diagnostics for acute infections because, as the Global Sepsis Alliance has acknowledged, COVID-19 can lead to sepsis—a life-threatening organ dysfunction caused by a dysregulated host response to infection. In severe COVID-19, sepsis looks like massive and systemic inflammation that attacks the organs. By taking advantage of this large change, it may be possible to examine the patient immune response and use it to diagnose severe COVID-19 and guide decision-making.
Immune response diagnostics
In fact, researchers are currently exploring different approaches to immune response diagnostics, which may be coming to the US for clinical use this year. Immune or “host-response” diagnostics overcome the limitations of current pathogen testing because they can detect any infection anywhere in the body by “reading” patterns and levels of immune system-related mRNAs from the blood. In other words, host-response diagnostics seek to leverage the body’s immune response to explain the cause of a patient’s symptoms.
mRNA-based immune response diagnostics seek to quickly answer three critical questions:
- Is infection present?
- Is it bacterial or viral?
- How severe will it get (based on predicted 30-day mortality)?
In a recent preprint published on MedRxiv, a 29-mRNA panel and algorithm focused on answering these three questions outperformed procalcitonin (PCT) and C-reactive protein (CRP) in distinguishing between bacterial and viral infections and noninfectious inflammation. This specific approach is unique because it was built using heterogeneous patient populations representing a variety of infections, ages, regions, and genders in order to be as clinically useful as possible across patients seen every day in emergency rooms. Other host-response approacheshave shown good results but in very narrow, homogeneous populations unrepresentative of daily practice.
In another preprint study on MedRxiv, researchers examined a set of six immune system-related mRNAs in multiple pre-COVID-19 cohorts, as well as in two prospective COVID-19 emergency department cohorts, to predict acute viral severity. The 6-mRNA signature was more accurate than several single biomarkers, including IL-6, PCT, CRP, lactate, and suPAR, in identifying patients who later developed respiratory failure or died. It is interesting and encouraging that a single small panel is able to predict outcomes in COVID-19 and non-COVID-19 viral infections alike, suggesting a durable pandemic product for years to come.
In triage situations that hospitals face every day, whether for “normal” acute infections and sepsis or for COVID-19, actionability is key. Novel, rapid host-response diagnostics are poised to help physicians make better hospital admission and resource decisions early in a patient’s course of treatment. Armed with this enhanced information, doctors will be able to more confidently make rule-in and rule-out decisions that will benefit these patients and the health care system that seeks to help them.
Timothy E. Sweeney, M.D., Ph.D., is cofounder and chief executive officer of Inflammatix, Inc. Inflammatix is a molecular diagnostics company building rapid, novel host-response tests for acute infectious and inflammatory conditions. Dr. Sweeney trained in general surgery at Stanford before cofounding Inflammatix. He can be reached at [email protected]
Originally published by Clinical Lab Manager, February 2021. Read online.